“Phytoplankton produce 50% of the oxygen we breathe” is a sentence we often read, particularly in media pieces destined to the general public. It makes for a poignant introduction and it highlights the crucial role of phytoplankton on planet Earth. It has been employed by several respectable (and respected) oceanographers: you can find it in this book review in Science, or here (with a focus on diatoms), in Le Monde, a famous french newspaper.
But there is a problem with this sentence: it’s false. Not because of nitpicking like “actually it’s only 46%”; it’s false by a very large margin. In this blog post, I’m going to try to explain why1, in the hope that scientists stop sharing this falsehood (though, given my modest readership, i expect the problem to persist for a bit.)
50% of primary production on Earth
If it seems so difficult to get rid of this false assertion, it’s because there is some kernel of truth in it.
Phytoplankton are responsible for half of the annual primary production on planet Earth2. In other words: 50% of the oxygen that is produced by photosynthesis every year on planet Earth comes from phytoplankton. The other 50% come from land plants. Macroalgae, marine angiosperms and microphytobenthos also produce O2 through photosynthesis but at the global scale their contribution is so small it can be neglected (or at least this is what i understood from Field et al.’s paper.)
Stocks and fluxes
The first problem with the sentence comes from a confusion between stocks and fluxes. Phytoplankton represent 50% of the annual production of O2, but we breathe from the atmospheric stock. Here is a plot representing the total mass of O2 in the atmosphere compared to the annual fluxes of O2 (positive and negative). Both are expressed in Gt of O2 (one gigaton = 1012 kg). The data come from this blog post (and sources therein) and from Huang et al. (2018).

Well, this is humbling. The entire activity of life on Earth for one year is contained in this minuscule brown dot labeled “Annual fluxes”. What this means is that when we breathe, we mainly sip from the vast atmospheric stock of O2.
This stock accumulated gradually for eons, since the apparition of the first photosynthetic organisms on our planet (estimated to have taken place 2.7-3.5 billion years ago)3. The vast majority of O2 molecules produced by photosynthesis were consumed by living organisms through respiration, the reverse reaction to photosynthesis. Respiration consumes organic matter and O2 to generate CO2 and energy. But a minority of organic matter was not consumed by respiration (nor by other non-living processes such as wildfires), and was trapped in rocks in the form of fossils, oil or coal. Because of this small imbalance between the mass of oxygen produced by photosynthesis and that which is consumed by respiration and non-living processes, over a very long time, O2 levels started to rise first in the early oceans, and then in the atmosphere, fueling the atmospheric stock of oxygen.
Long story short, the oxygen we consume was mainly produced by photosynthesis reactions that took place over billions of years during the ancient times of life on Earth. Another (strange) way to say it: we are breathing the waste of our great-great-great-great-great-great-[…]-great-grandparents’ vegetables.
What happens in the ocean stays in the ocean
If this “stocks and fluxes” argument didn’t convince you already, I have another card up my sleeve. Let’s look at the tiny brown dot in more detail (data from Huang et al., 2018, figure 4).
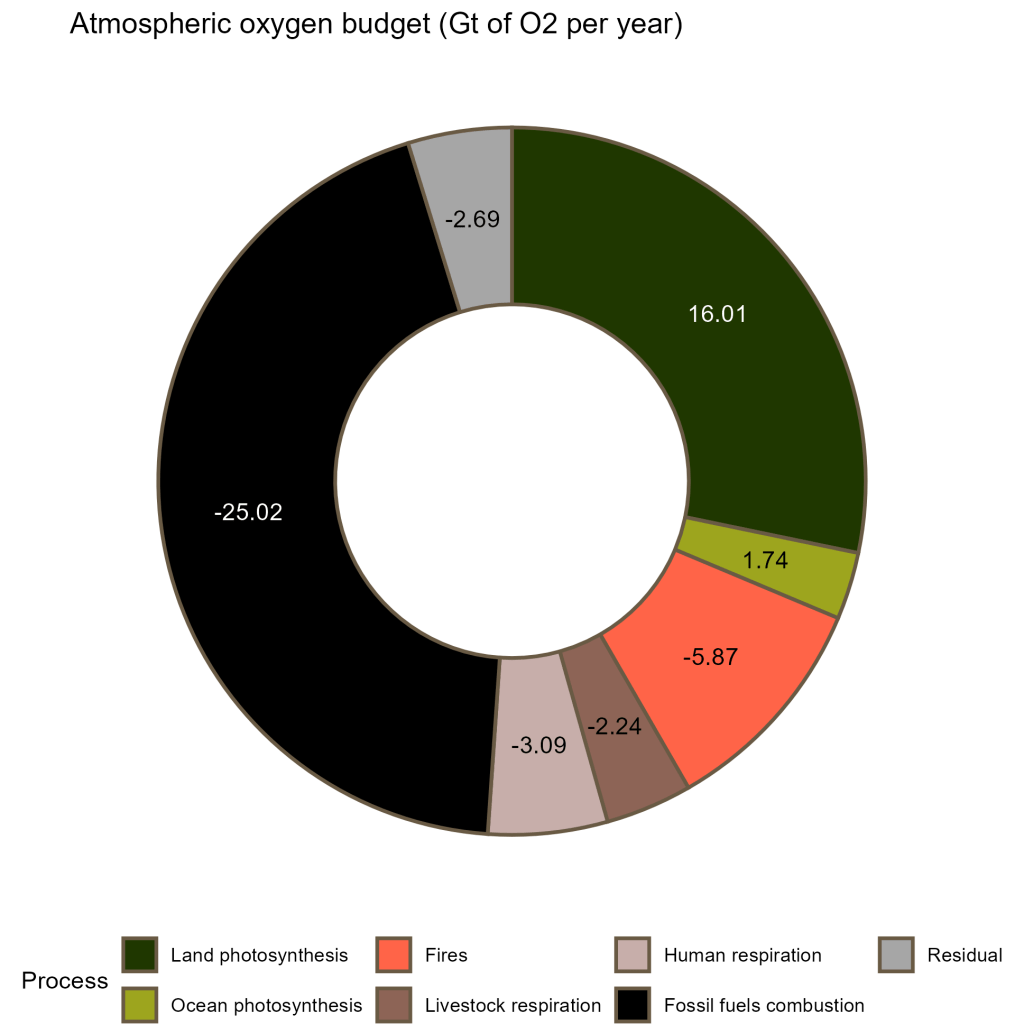
This doughnut plot represents the annual atmospheric oxygen budget: the fluxes of oxygen that go in (“sources”, positive numbers) and out (“sinks”, negative numbers) of the atmospheric stock, due to various processes. There are 2 main sources: photosynthesis by land organisms (+ 16.01 Gt of O2 per year) and photosynthesis by ocean organisms (+ 1.74 Gt of O2 per year).
But if phytoplankton are responsible for half of the Earth’s photosynthetic activity, how is it possible that its contribution is only 1/9th that of land plants?
This is because the oxygen produced by phytoplankton is not released directly in the atmosphere, but in seawater where the cells live. While dissolved in the water, O2 molecules are consumed by the respiration of all the heterotrophic organisms of the sea: fish, jellyfish, zooplankton, crabs, bacteria… (and also by phytoplankton themselves, at night!)
The vast majority of the oxygen produced by phytoplankton is therefore consumed directly in the ocean, and remains unaccessible to humans (unless you happen to have gills.) Only a small proportion “escapes” into the air by diffusion at the surface of the ocean.
I think this justifies saying farewell to our old friend “Phytoplankton produce half of the oxygen we breathe”. It was good while it lasted.
The decrease in atmospheric oxygen
If you looked closely at the second plot, you might have noticed something: oxygen sinks outweigh sources. Nowadays, there is not enough new oxygen coming into the atmosphere to compensate what is burned out. This is mainly due to human consumption of fossil fuels: we are essentially burning the fossil reserves of organic matter that accumulated for billions of years, consuming atmospheric oxygen and releasing carbon dioxide in the process.
For several decades, the oxygen in the atmosphere has been decreasing at the same time CO2 concentrations have been rising. This has been measured very nicely by the O2 program of the Scripps Institution of Oceanography. But we should not worry too much about oxygen depletion: because the O2 fluxes are so small compared to the atmospheric stock, there is a multitude of other problems (global warming, biodiversity loss, antimicrobial resistance, pick your favourite!) that we will have to deal with much, much sooner!
This blog post and all the figures it contains were produced by me, and are under a Creative Commons Attribution (CC BY) licence. You can reuse it freely as long as you cite the author (Victor Pochic) properly. All the R scripts and data files necessary to reproduce the figures are available on my github.
- The NOAA has a nice concise page on its website on roughly the same subject, if you don’t want to read through the rantings of an obscure french blogger. ↩︎
- Field et al. (1998) ↩︎
- Most of my understanding of the history of oxygen on Earth comes from the book Oxygen, the molecule that made the world by Nick Lane. Many thanks to my colleague Rossana for lending me her copy! ↩︎

Leave a comment