This is the second part of my series on Dinophysis. If you didn’t read the first part, go read it, and come back!
Taming the beast
After the toxicity of Dinophysis was revealed, the incentives for establishing permanent cultures in laboratory conditions were high. Once mastered, lab cultures of microorganisms are a great asset for future investigations: setting up controlled, ambitious experiments is much easier when you are guaranteed a steady supply of relatively similar subjects, instead of relying on the often unpredictable fluctuations of heterogenous natural populations.
Cultivating microalgae in a lab is, in theory, relatively simple. All you need is clean seawater with a supplement of nutrients (nitrogen and phosphate, mostly) and some white light. Pick a cell of interest from a natural sample, put it in culture medium, and let photosynthesis and asexual reproduction work their magic. Dinophysis, however, stubbornly resisted all cultivation attempts. Isolated cells would survive for several weeks in glassware, sure, but would-be Dinophysis breeders could not yield any significant growth, and would ultimately watch their cultures reach a heartbraking and systematic demise.
Let’s talk about the Anna Karenina principle.
…
Leo Tolstoi’s novel Anna Karenina begins with this sentence:
Happy families are all alike; every unhappy family is unhappy in its own way.
From this famous incipit, a theorem emerges: for a family to be happy, every condition of happiness must be fulfilled. A failure to meet any of the conditions will lead to unhappiness. Thus, two families can be equally unhappy for wildly different reasons. Substitute family with any human enterprise, and happiness with successful outcome, and you get the Anna Karenina principle.
In his book Guns, Germs and Steel (Chapter 9), the biologist and ecologist Jared Diamond applies the Anna Karenina principle to the domestication of animals by humans. For a process of domestication to be successful, the candidate animal species needs a “perfect match” of characteristics that make its domestication possible.
Among other requirements, Diamond explains that the animal must be relatively easy to feed: a picky animal that only eats very specific things makes a poor candidate for domestication. Also, the animal must tolerate social life within or near human groups, and with its conspecifics. A failure in the first condition will prevent domestication, regardless of how well the animal suits the second condition, and vice-versa. For example: the placid, sociable koala seems to conform perfectly to domestic life1, but it is so picky in its diet (it’s eucalyptus or nothing) that it is a pain to feed. Conversely, a bear will eat more or less any junk that comes its way, but its solitary and territorial lifestyle (plus its tendency to dismember humans at the slightest annoyance) probably prevented it from becoming Man’s best friend.
…
The Anna Karenina principle explains why the most well-studied organisms are also, arguably, the ones that are most easily kept and bred in a lab. Most of what we know in biology comes from studies on a few such species, called model organisms. A few well-known examples are the bacterium Escherichia coli, the nematode worm Caenorhabditis elegans, the plant Arabidopsis thaliana or the proverbial lab mouse.
For microalgae, the most studied model organism is, I think, the diatom Phaeodactylum tricornutum. Many researchers have conducted experiments on this species to understand the inner workings of photosynthesis at the physiological and molecular levels (Croteau et al., 2025). Mutants of P. tricornutum are often created to elucidate the functions of specific genes (Fortunato et al., 2016). Phaeodactylum tricornutum is a remarkable organism. As my friend Thomas likes to say, it is “the lab mouse of microalgae”2. The problem is: if you want to study something as strange as a platypus, chances are your setup for breeding lab mice will not cut it.
I hope you’re not lost after this long rambling about domestication and model species. The key idea here is that Dinophysis turned out to be a platypus.

The mistery of orange plastids
Faced with recalcitrant Dinophysis cultures, researchers did… well, research. In 1988, Schnepf and Elbrächter published an analysis of the structure and fluorescence of the plastids found in Dinophysis cells. Their main finding was that the plastids of Dinophysis, contrary to those of other dinoflagellates, emitted orange fluorescence, betraying the presence of a specific pigment : phycoerythrin. More specifically, several structural elements of the plastids, revealed by electron microscopy, showed great similarity with plastids of cryptophytes (another family of small microalgae). Such features are seldom observed in photosynthetic dinoflagellates.
The same year (1988), Ishimaru and colleagues observed that the orange fluorescence of cultivated cells of Dinophysis decreased gradually during their survival in the lab. Fluorescence decreased less, and cells seemed to survive longer, when Dinophysis was cultivated with the co-occurring cryptophyte Plagioselmis. The researchers even claimed to have observed ingestion of Plagioselmis cells by Dinophysis. For the first time, they formulated a key hypothesis: that the chloroplasts of Dinophysis might not be its own, and that it stole them from a prey. This biological strategy is known as kleptoplastidy.
Alas, even Dinophysis cells supplied with cryptophyte prey did not last past a few months in the lab, and they could not divide into a sustainable culture. Either the kleptoplastidy hypothesis was false, or something was missing.
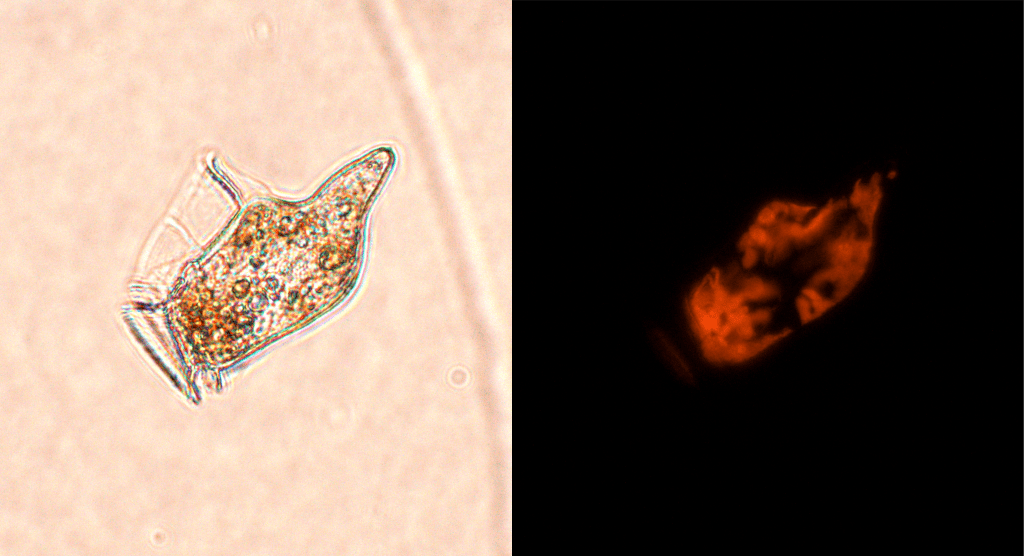
The missing link
Dinophysis was not the only planktonic microorganism in which the strange orange plastids had been observed. A ciliate named Mesodinium rubrum also harboured such chloroplasts. Large, dark red blooms of this unicellular organism in marine waters had been documented at least since Darwin’s voyage on the Beagle3, and its photosynthetic productivity rivaled that of the most productive phytoplankton. However, it had been overlooked by most phytoplankton surveys, on the basis that it was – taxonomically-speaking (as a ciliate) – a member of the microzooplankton. In his great review article (the aptly titled Mesodinium rubrum: the phytoplankter that wasn’t), David Crawford of Southampton University attempted to call attention to this neglected phytoplankter. One author he cites (Taylor, 19824) describes “the association between M. rubrum and its cryptomonad symbiont to be the most strongly integrated known, between host and photosynthetic cytobiont.” Crawford’s paper and subsequent work on Mesodinium rubrum have probably contributed to ignite (or reinforce) the interest of other scientists in this peculiar photosynthetic ciliate. One thing is for sure, his 1988 review was cited 12 years later in an article that would contribute to shift our perspective on how kleptoplastidic microorganisms operate.
In 2000, a team of biologists at the university of Maryland published their striking finding: their antarctic strain of Mesodinium rubrum grew by stealing not only the plastids, but also the nucleus of its cryptophyte prey. The biological implications were huge5. But for those who had been trying relentlessly to cultivate Dinophysis, the biggest news was that they had a new thing they could try to feed their platypus. Soon enough, a genetic analysis revealed that plastids inside the cells of Dinophysis were identical to those of Mesodinium’s cryptophyte prey. Sven Janson, author of this discovery, wrote the following, that now reads like a prophecy of stunning accuracy:
“One possibility that has not been pointed out previously is that the plastids could enter Dinophysis spp. from M. rubrum as secondary kleptoplastids. After all, Dinophysis spp. have been shown to feed on ciliates, whereas consumption of cryptophytes remains to be proven (Hansen, 1991). Keeping in mind that all attempts to cultivate Dinophysis spp. have failed so far, an unexpected source and need of plastid may be a reason for this and feeding experiments involving M. rubrum are highly interesting in this respect.”
Finally, in late 2005 (25 years after the reveal of its toxicity!), the first lab culture of Dinophysis was established, in South Korea. Park Myung Gil and his colleagues described how they achieved the cultivation of Dinophysis in a 3-level, lab-controlled trophic chain: the cryptophyte Teleaulax sp. was fed to Mesodinium rubrum6, which was in turn fed to Dinophysis acuminata. Park et al. (2006) is a truly great article. On top of achieving a real breakthrough (the end of a 25-year puzzle for a lot of marine biologists!), it also comes with lots of photos, and even a video of Dinophysis feeding on its prey.
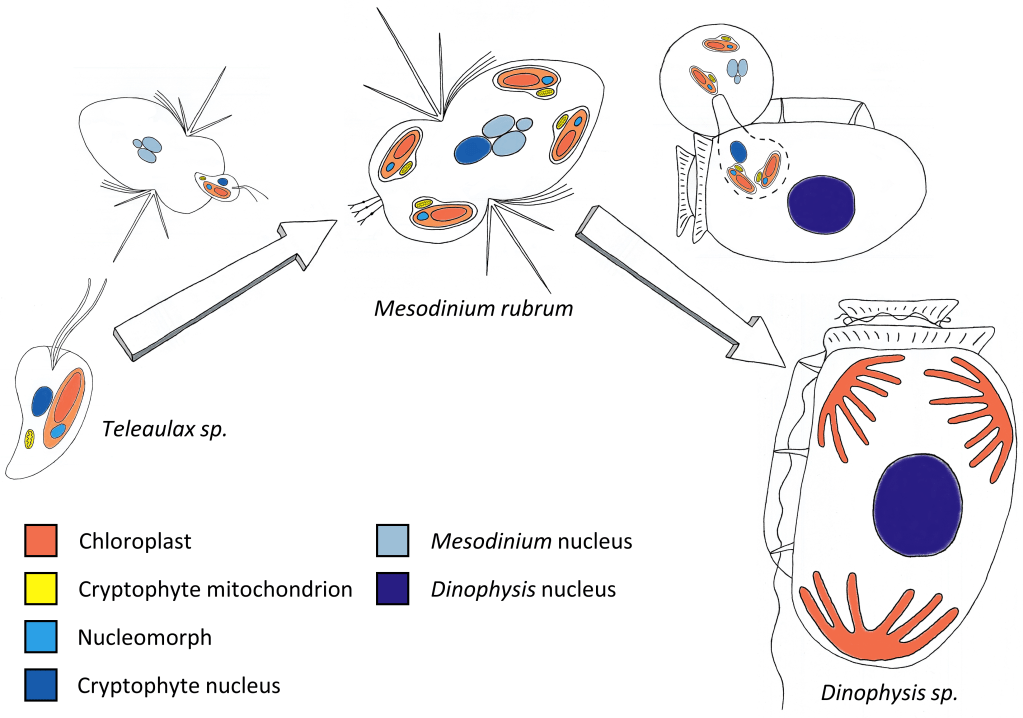
Nowadays, Dinophysis strains from the coasts of America, Europe, Japan and elsewhere are cultivated in research labs, with the same system Park and his colleagues presented in their paper. This is still a challenging task (remember we’re beating the Anna Karenina principle here!), that requires expertise and dedication, and only a handful of institutes in the world keep permanent cultures7. These cultures are invaluable for studying the ecophysiology of a hazardous toxic dinoflagellate. And, from a more “basic research” standpoint, they also provide not one but two incredible models for studying kleptoplastidy and the acquisition of photosynthesis in unicellular plankton.
This post is the second in my 3-part series on Dinophysis.
Here are all the references for the sources of this (very long) post:
- Anna Karenina principle and domestication:
Diamond, Jared. Guns, Germs and Steel, the fate of human societies (1997, first edition), Chapter 9 “Zebras, unhappy marriages, and the Anna Karenina principle”
- Difficulties of first cultures of Dinophysis:
Ishimaru T., et al. “Cultures of Dinophysis fortii and D. acuminata with the cryptomonad, Plagioselmis sp.” JSM Mycotoxins (1988) https://doi.org/10.2520/myco1975.1988.1Supplement_19
Maestrini Serge Y, et al. “Dinophysis spp. cells concentrated from nature for experimental purposes, using size fractionation and reverse migration.” Aquatic Microbial Ecology. 9 (2). 177-182. (1995). https://doi.org/10.3354/ame009177, https://archimer.ifremer.fr/doc/00258/36956/
- Strange cryptophyte plastids in Dinophysis:
Schnepf, E. and Elbrächter, M. “Cryptophycean‐like double membrane‐bound chloroplast in the dinoflagellate, Dinophysis Ehrenb.: evolutionary, phylogenetic and toxicological implications.” Botanica Acta 101.2 (1988): 196-203. https://doi.org/10.1111/j.1438-8677.1988.tb00033.x
Janson, Sven. “Molecular evidence that plastids in the toxin‐producing dinoflagellate genus Dinophysis originate from the free‐living cryptophyte Teleaulax amphioxeia.” Environmental Microbiology 6.10 (2004): 1102-1106.
- Mesodinium rubrum and kleptoplastidy:
Crawford, David W. “Mesodinium rubrum: the phytoplankter that wasn’t.” Marine Ecology Progress Series 58 (1989): 161-174. https://www.jstor.org/stable/24842177
Gustafson, Daniel E. Jr., et al. “Cryptophyte algae are robbed of their organelles by the marine ciliate Mesodinium rubrum.” Nature 405.6790 (2000): 1049-1052. https://doi.org/10.1038/35016570
- First successful culture of Dinophysis:
Park, Myung Gil, et al. “First successful culture of the marine dinoflagellate Dinophysis acuminata.” Aquatic Microbial Ecology 45.2 (2006): 101-106. https://doi.org/10.3354/ame045101
…
Illustrations with no source indicated in the legend are personal productions by V. Pochic. You can reuse the content of this post freely as long as the author (and original works cited herein) are properly referenced (Creative Commons attribution license).
- This is a supposition, I confess I don’t know much about koalas. Apparently they can be quite scary. ↩︎
- Interestingly, the very characteristics that make Phaeodactylum so powerful as a model organism (i.e., easy to cultivate, high growth rate) are also those that somewhat set it apart from other phytoplankton species, and preclude hasty generalisations. For example, it is a diatom that lacks the characteristic silica cell wall of diatoms! In a recent study on phytoplankton from the Baltic sea (Orizar and Lewandowska, 2025), researchers observed that, contrary to most other phytoplankton that exhibit a clear salinity preferendum, P. tricornutum grows evenly regardless of the salinity. That thing just doesn’t care. ↩︎
- I will definitely write something about Darwin and plankton blooms some day. ↩︎
- I’ve tried to go to the original source, but the best thing I can find on Google Scholar is a citation. The “paper” version of the paper (published in the Annales of the Institut Océanographique de Paris) can probably be found in some library in Paris or in the marine stations in Villefranche or Roscoff? If you, reader, happen to be near these places and are brave enough to look for old scientific papers in a library (!!), I’d be grateful if you can send me a scan! ↩︎
- This will be the subject of the 3rd episode in the series. ↩︎
- Park et al. use the name Myrionecta rubra, a taxonomic synonym of Mesodinium rubrum that is now obsolete. ↩︎
- Luckily for me, it’s the case in Ifremer Nantes where I work! I have to say here my admiration for my colleagues Liliane and Véronique for all the energy they put into isolating and growing the many strains of Dinophysis that we can photograph and experiment on. ↩︎

Leave a comment