This is the third part of my series on Dinophysis. If you didn’t read the first and second part, go read them and come back!
We’ve now seen how Dinophysis was finally cultivated in the lab: by feeding it with its prey, the ciliate Mesodinium rubrum. Both of these microbes thrive by acquiring chloroplasts from foreign origin. This is kleptoplastidy.
We need to talk about kleptoplastidy.
Thieves!
Chloroplasts (or plastids) are cellular compartments within photosynthetic cells, within which the chemical reactions of photosynthesis take place1.
However, some heterotrophic (non-photosynthetic) organisms that prey on photosynthetic cells also harbour chloroplasts. After ingesting their prey, they do not digest the chloroplasts – at least not immediately. They keep these foreign organelles alive within their own cells, and the stolen plastids (or kleptoplastids) keep photosynthesising for a while within their new home.
Are kleptoplastidic organisms rare? Well, yes and no. On one hand, you’ve never seen a terrestrial herbivore turn green after eating enough grass. On the other hand, if you look closely enough into a meadow of green algae in shallow waters, you might find yourself face to mantle with this pretty thing:

Sea slugs in the genus Elysia are well-known examples of kleptoplastidic animals. They feed on algae, but instead of digesting the plastids they carry them around just under their skin, where photons from the sunlight can be harvested by the photosystems, turned into chemical energy and used to produce organic molecules.

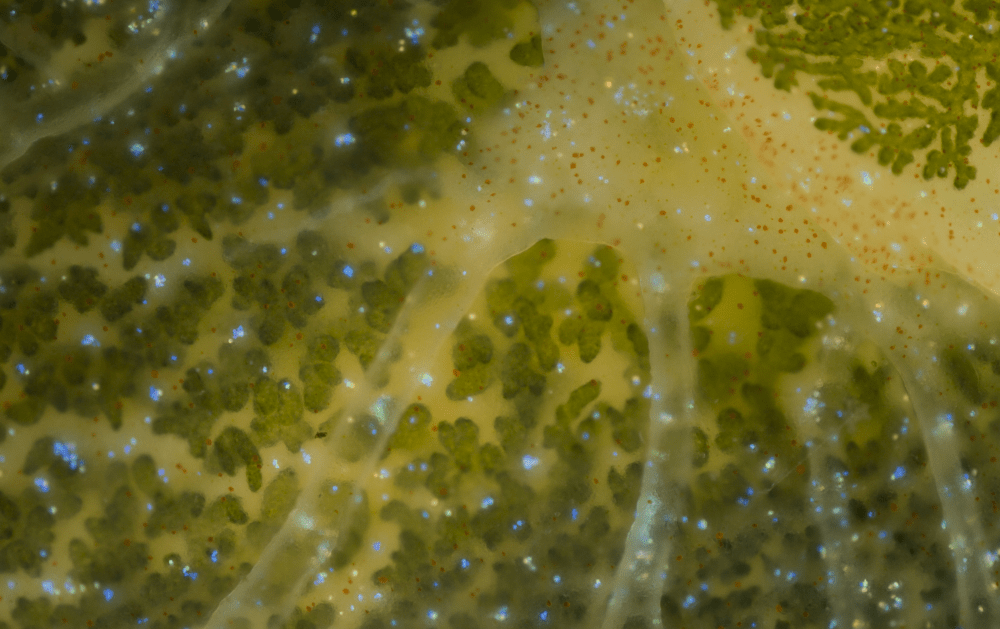
The slug can use the molecules produced by the kleptoplastids as a complementary source of organic matter, on top of its grazing2. Also, when food becomes scarce, kleptoplastids continue to photosynthesise and provide the slug with energy in the form of starch, allowing it to withstand prolonged periods of starvation3. They cannot survive like this forever, though. Eventually, the jig is up, the kleptoplastids decay and end up digested. The slug must feed regularly on algae to refurbish its verdant coat. But why? Plants never need to acquire new chloroplasts, so what’s the difference?
Control over the garden
In fully photosynthetic organisms, chloroplasts divide when the mother cell divides, and are thus passed on to daughter cells indefinitely. In kleptoplastidic organisms, it doesn’t really work like this.
Let’s turn back to the microbial world, and to our ciliate friend Mesodinium rubrum. Mesodinium eats specific cryptophyte microalgae, and keeps their plastids intact within its cytoplasm. But it also keeps the nucleus. And that’s rather unusual. And important.
See, a chloroplast is a part of a cell. It cannot survive by itself. When some parts of the chloroplast (membrane lipids, photosystem proteins, pigments…) are inevitably damaged by light, they need to be replaced with new ones. This necessitates the synthesis of new proteins, that are encoded by genes. The chloroplast harbours a modest “within-house” genome, but a lot of the genetic sequences necessary for its maintenance are actually in the nuclear genome.
Think of the chloroplast like a vegetable garden. You can steal a garden, this will give you food for a while. But if you want to maintain it properly and exploit it to its full potential, you also need to steal the tool shed. That’s what Mesodinium does, it steals the tool shed.
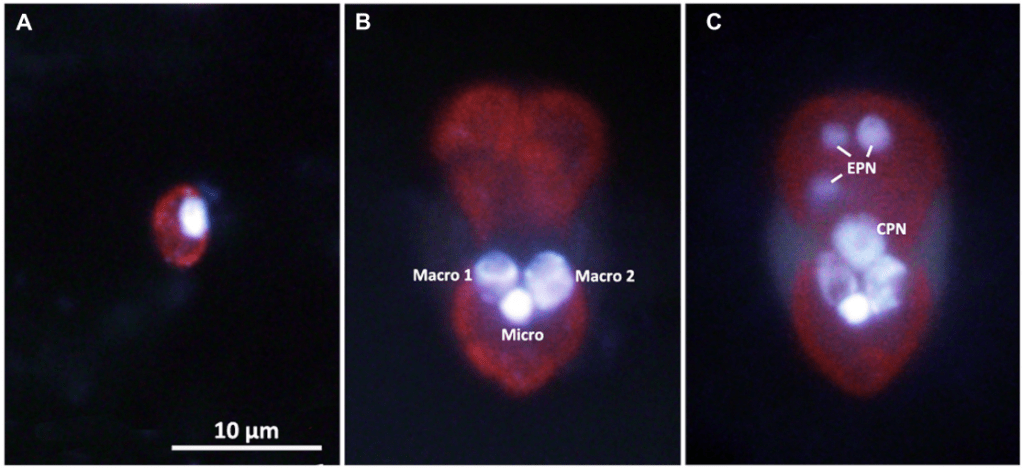
As you can see in panel C of the figure above, the stolen cryptophyte nucleus (or kleptokaryon) is “plugged” near the ciliate’s own nuclei. Once there, it is put to good use with intense transcription, helping the ciliate to maintain and multiply its plastids4. In fact, the hijacked nucleus turns Mesodinium into a very productive photosynthesizer.
(By the way, this nucleus hijacking is an incredible biological feat. Even more so when you consider that ciliates have a different genetic code. This creature is able to transcribe and translate 2 nuclear genomes with 2 different genetic codes at the same time!)

Still, while the stolen nucleus allows to keep the kleptoplastids afloat, a new problem arises at the time of cell division. The kleptocaryon’s genome cannot be replicated and thus one daughter cell is always going to get wronged and will need to acquire a new kleptokaryon by itself if it wants to maintain its chloroplasts in working order5.
Also, one kleptokaryon cannot be used forever. After some time, it becomes non-functional and is presumably digested or decommissioned one way or another. So, although it may seem a skilled phytoplankton in the right conditions, Mesodinium is always dependent on an intake of fresh cryptophyte prey. As Johnson and colleagues poetically put it, it is “caught in a Promethean struggle of perpetually needing to reacquire the kleptokaryon.”
Of proteins and genes
Dinophysis preys on Mesodinium, and so relies on the same kleptoplastids of cryptophyte origin. However, it does not retain the cryptophyte nucleus. What happens when you try to maintain the garden without the proper tool shed?
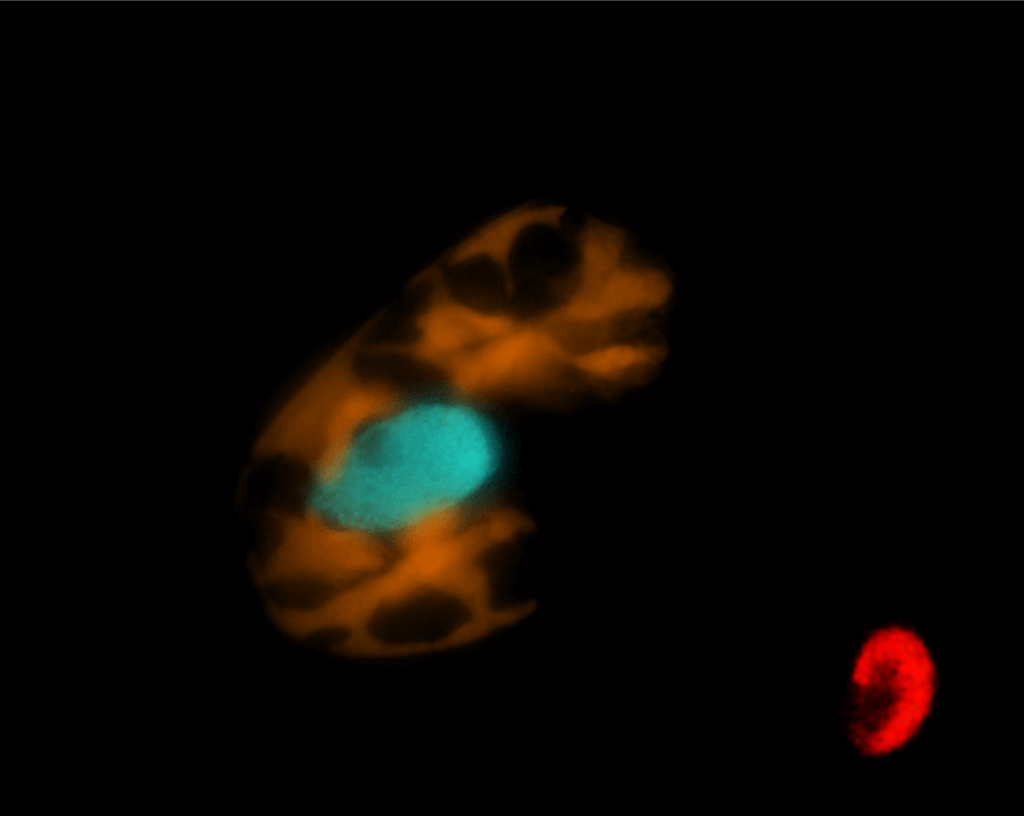
Dinophysis can grow photosynthetically for approximately 2 weeks (in experimental conditions) off the back of its kleptoplastids. After that, without fresh prey, its growth gets halted. This has to do with the fact that kleptoplastids become less and less functional as time goes by, because accumulated damage cannot be repaired.
A very nice example of this has to do with phycoerythrin (PE), the protein that gives cryptophyte plastids their distinctive orange fluorescence. Phycoerythrin is an essential component of their photosynthetic machinery.
The cryptophyte phycoerythrin is actually a protein complex made of 2 different subunits: the alpha- (α) and bêta- (β) subunits. 2 α-subunits and 2 β-subunits assemble to form a molecule of cryptophyte phycoerythrin:
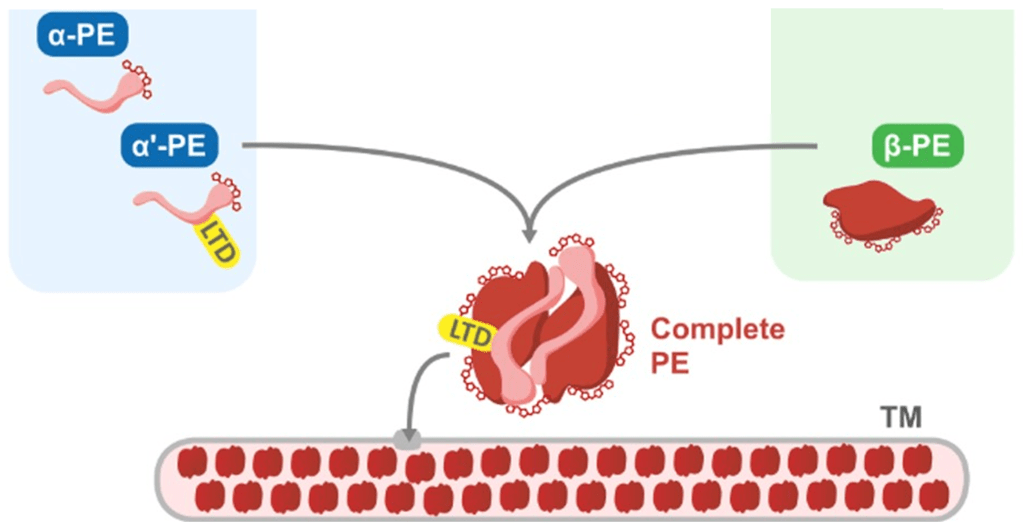
The assembly of the subunits takes place inside the chloroplast. Thanks to the Luminal Targeting Domain (LTD) bound to the α-subunit, the complete cryptophyte PE is transferred to the thylakoid lumen, a sub-compartment within the chloroplast, where it can do its photosynthetic job of harvesting green light.
But there’s a catch here: while the β-subunit is encoded by a gene in the chloroplast’s own genome, the α-subunit is encoded by a gene in the nuclear genome. The α-subunit is synthesised outside of the chloroplast, and is then sent there to be assembled into the complete PE-complex. So what happens when Dinophysis, who lacks the cryptophyte nucleus, tries to produce a cryptophyte phycoerythrin?
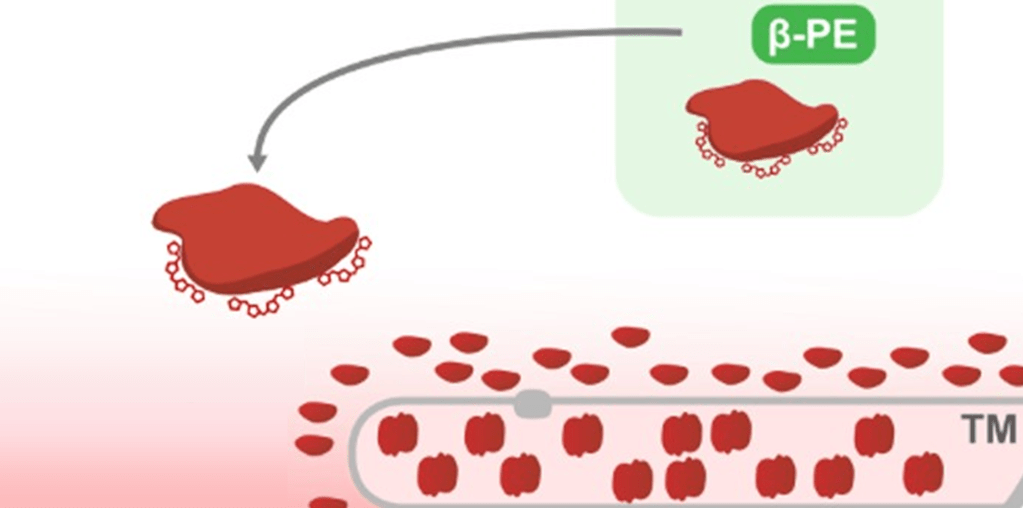
Dinophysis synthesises an incomplete version of the cryptophyte PE, formed of lonely β-subunits. Also, because it lacks the LTD, it probably lingers pitifully around the thylakoid membrane, unable to enter the lumen.6
This is but one example of the importance of the nuclear genome for the maintenance of plastids, as many essential chloroplastic proteins are encoded by nuclear genes.
So here are our planktonic sorcerer’s apprentices, sloppily mimicking the magic of photosynthesis. But is this the end of the story? Actually, I’ve not told you everything about phycoerythrin…
The long path towards photosynthesis
Let’s look closely at those phycoerythrins.
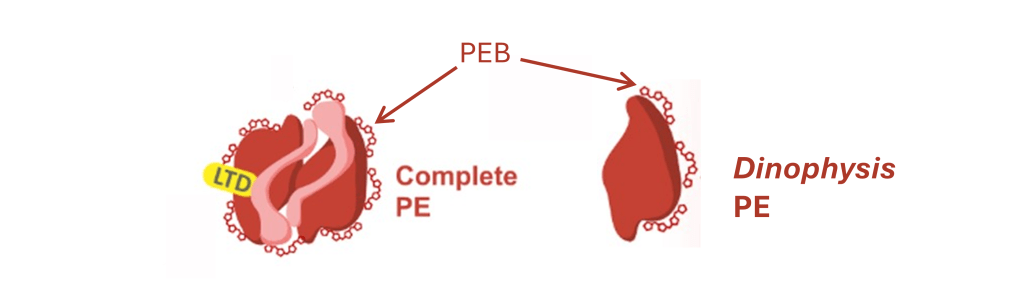
These little molecules at the surface of the PE-subunits are pigments called phycoerythrobilins (PEB). These pigments are essential to the absorption of light by the PE molecule. Their synthesis necessitates enzymes encoded by genes from the cryptophyte nuclear genome, called pebA and pebB. So how is it possible that they are also found in the Dinophysis-produced PE?!
Maybe you guessed it by now, the nuclear genome of Dinophysis contains pebA and pebB genes of cryptophyte origin, and the cell uses them to synthesise PEB. They were copied at some point in the past from the genome of the cryptophyte nucleus through a process called horizontal gene transfer. In fact, it is not uncommon to find some genes essential for the maintenance of chloroplasts in the nuclear genome of kleptoplastidic dinoflagellates7.
What this means is that, over a very long time and repeated interactions between kleptoplastidic organisms and their prey, many gene transfers can take place. Gene transfers that enable predators to better control their kleptoplastids will confer an advantage to these cells, and be selected in the population. In the long run, after enough gene transfers from the prey to the predator, the kleptoplastids can be maintained indefinitely. The predator becomes a host, the kleptoplastid becomes a chloroplast. The kleptoplastidic interaction turns into a permanent endosymbiosis.
As you may know, the endosymbiosis of a cyanobacteria by an ancient eukaryote is the event that led to the evolution of the archaeplastids, the vast family encompassing red and green algae, and all of land plants. And, after secondary and tertiary endosymbioses, of all the photosynthetic eukaryotes we know of today.

This is part of the beauty of studying kleptoplastidic organisms: as an intermediate stage between heterotrophs and fully photosynthetic organisms, they offer us a peek into how the first photosynthetic eukaryotes may have lived, the hurdles they had to overcome. They allow us to learn about one of the most decisive events in the history of life on Earth.
…
The idea of kleptoplastidy being a stepping stone towards permanent, autonomous photosynthesis is appealing, quite widespread, and probably true in a broad way. The story of the “sorcerer’s apprentices” on their evolutionary way to mastering photosynthesis is a compelling and engaging one (that’s, obviously, what prompted me to write this blog post). However, I also feel that ending the story this way would be unfair to Elysia, Mesodinium, Dinophysis and all the other weird plastid thieves out there.
Their kleptoplastidic strategy is not merely a step towards a goal, a burdensome and clumsy endeavour that needs to lead to permanent endosymbiosis to be considered successful, or an evolutionary dead end if it doesn’t. That’s not how evolution operates. Kleptoplastidy is a surprising and yet effective lifestyle that has emerged multiple times across the tree of life, because it just works, right now, regardless of what may or may not happen in a distant future.
- Actually, there are photosynthetic cells without plastids: cyanobacteria. I wonder if they may be relevant to this story at some point… ↩︎
- Cartaxana et al. (2021) ↩︎
- Laetz et al. (2017) ↩︎
- Altenburger et al. (2021) ↩︎
- As seen in panel C of the figure from Kim et al., one Mesodinium cell can accumulate extra cryptophyte nuclei (EPN in the figure) if it catches more than one crypto. These extra nuclei can be shared between daughter cells, but you see how this just merely pushes the problem a few generations further. ↩︎
- This model for the Dinophysis-produced phycoerythrin is detailed in the very nice paper of Garric et al. (2024), where substantial experimental evidence is presented in support of it. I can’t go into details here, but if you’re interested go check the paper or chapter 2 of Sarah Garric’s PhD thesis (which is open access). ↩︎
- Hehenberger et al. (2017) ↩︎
This was the final episode in my 3-part Dinophysis series!
Many many thanks to my PhD supervisor Bruno Jesus for allowing me to use his wonderful photos of Elysia viridis to illustrate this post.
The text in this blog post is a personal production of V. Pochic and is under a CC BY 4.0 license, as are the images for which credit is not indicated in the legend.
Credit for the images in the “diversity of photosynthetic eukaryotes” illustration:
(From left to right and from top to bottom)
- Leaf tip of Elodea canadensis, CC BY KarlGaff
- Pinus strobus foliage, CC BY Johndan Johnson-Eilola
- Ceramium, red algae, CC BY Ivan Bachev
- Coscinodiscus diatom, CC BY Victor Pochic
- Bryum argenteum, moss, CC BY Paul van de Velde
- Yellow flowers of Senna sp., CC BY Victor Pochic
- Glaucocystis sp., glaucophyte, CC BY Neon_ja
- Helianthemum apenninum flowers, CC BY Victor Pochic
- Young sporophyte of Splachnum ampullaceum, CC BY Des_Callaghan
- Acer palmatum, japanese maple, CC BY Protolanguage17
- Pulsatilla alpina, alpine anemone, CC BY Victor Pochic
- Lepidodinium chlorophorum, dinoflagellate, CC BY Victor Pochic
- Nymphaea alba, CC BY Victor Pochic
- Kelp forest in Channel Islands, California, CC BY Aquaimages
- Bristlecone pines in White mountains, California, CC BY Rick Goldwaser
- Timema poppense (this one is not photosynthetic!) camouflaged on its host Sequoia sempervirens, CC BY Moritz Muschick (University of Sheffield)
Bibliography:
Cartaxana, Paulo, et al. “Photosynthesis from stolen chloroplasts can support sea slug reproductive fitness.” Proceedings of the Royal Society B 288.1959 (2021): 20211779.
Laetz, Elise MJ, et al. “Photosynthate accumulation in solar-powered sea slugs-starving slugs survive due to accumulated starch reserves.” Frontiers in zoology 14.1 (2017): 4.
Kim, Miran, et al. “Dynamics of sequestered cryptophyte nuclei in Mesodinium rubrum during starvation and refeeding.” Frontiers in microbiology 8 (2017): 423.
Altenburger, Andreas, et al. “Limits to the cellular control of sequestered cryptophyte prey in the marine ciliate Mesodinium rubrum.” The ISME Journal 15.4 (2021): 1056-1072.
Johnson, Matthew D., et al. “Functional control and metabolic integration of stolen organelles in a photosynthetic ciliate.” Current Biology 33.5 (2023): 973-980.
Garric, Sarah, et al. “Impaired photoacclimation in a kleptoplastidic dinoflagellate reveals physiological limits of early stages of endosymbiosis.” Current Biology 34.14 (2024): 3064-3076.
Hehenberger, Elisabeth, Rebecca J. Gast, and Patrick J. Keeling. “A kleptoplastidic dinoflagellate and the tipping point between transient and fully integrated plastid endosymbiosis.” Proceedings of the National Academy of Sciences 116.36 (2019): 17934-17942.

Leave a comment